Abstract
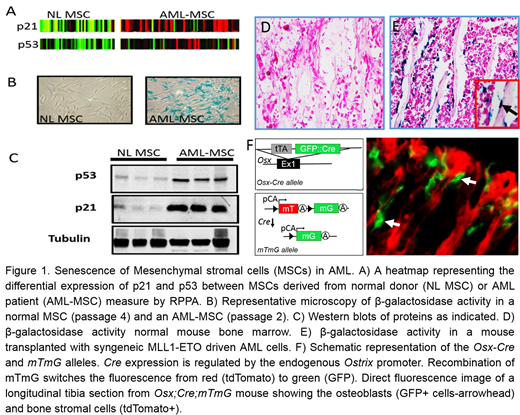
Acute myeloid leukemia (AML) is a genetically heterogeneous malignancy characterized by bone marrow infiltration of abnormally proliferating leukemic blasts which results in fatal anemia, bleeding and infectious complications due to compromised normal hematopoiesis. Patients with complete remission (CR) but incomplete blood cell count recovery (CRi) have significantly shorter survival compared to CR patients. Although there is a correlation between CRi and minimal residual disease (MRD), the two variables were shown to be independent risk factors for relapse development (1). The mechanism by which AML induces bone marrow failure in patients is largely unknown. Here, we demonstrate that AML derived MSCs highly express p53 and p21 proteins and are more senescent compared to their normal age-matched controls as demonstrated by high β-galactosidase staining (figure 1. A, B&C). Emerging evidence indicates that the aging of endosteal niche cells results in lower reconstitution potential of hematopoietic stem cells (2). To functionally evaluate the effects of AML on bone marrow MSCs, we utilized a murine leukemia model of the AML microenvironment. We transplanted Osx-Cre;mTmG mice with AML cells and compared the senescence of MSCs in normal bone marrow (Figure 1.D) with AML (Figure 1.E). Consistent with our initial findings in human, AML strongly induced senescence of osteoblasts. This suggests that AML suppresses normal hematopoiesis by inducing senescence in the hematopoietic niche. To address the role of p53 signaling in senescence of MSCs we generated a traceable conditional p53 gain/loss model specifically in bone marrow MSCs using Osx-Cre;mTmG; Mdm2fl/+ and Osx-Cre;mTmG;p53fl/fl mice respectively (Figure 1.F). Deletion of p53 in bone marrow MSCs resulted in an increased population of osteoblasts (GFP+) in Osx-Cre;mTmG;p53fl/fl mice in comparison to Osx-Cre;mTmG mice suggesting that p53 loss in osteoblasts inhibits senescence of osteoblasts. In order to evaluate p53 activity after recombination of p53fl alleles in the osteoblasts, we isolated MSCs from bone marrows and analyzed the expression of p21.P21 was significantly down regulated in osteoblasts (GFP+) derived from Osx-Cre;mTmG;p53fl/fl mice whereas its expression in the hematopoietic cells from same tissue (tdTomato+) remained comparable to p53 wild type suggesting that p21 as the master regulator of senescence is regulated by p53 in bone marrow mesenchymal cells. To evaluate the effect of p53 loss in osteoblasts and its impact on hematopoietic cells, we isolated the GFP+ cells (osteoblasts) and RFP + cells (hematopoietic) by FACS. Senescent cells, non-cell autonomously, modulate the bone marrow microenvironment through the senescence-associated secretory phenotype (SASP). We analyzed the expression of fifteen SASP cytokines by QPCR. Deletion of p53 in bone marrow mesenchymal cells strongly abrogated the expression of several SASP cytokines. Interestingly several Notch target genes such as Hey1 and Hey2 were highly induced in MSCs following p53 deletion suggesting a role for Notch signaling in hematopoietic failure following AML induced MSCs senescence.
Our data suggest that AML induces senescence of endosteal niche resulting in hematopoietic failure. These findings contribute to our understanding of the role of p53 in leukemia MSCs and could have broad translational significance for the treatment of hematopoietic failure in patients with AML.
Chen X, et al. (2015) Relation of clinical response and minimal residual disease and their prognostic impact on outcome in acute myeloid leukemia. J Clin Oncol 33(11):1258-1264.
Li J, et al. (2018) Murine hematopoietic stem cell reconstitution potential is maintained by osteopontin during aging. Sci Rep 8(1):2833.
Andreeff:Astra Zeneca: Research Funding; Daiichi-Sankyo: Consultancy, Patents & Royalties: MDM2 inhibitor activity patent, Research Funding; United Therapeutics: Patents & Royalties: GD2 inhibition in breast cancer ; Celgene: Consultancy; Eutropics: Equity Ownership, Membership on an entity's Board of Directors or advisory committees; Amgen: Consultancy, Research Funding; Oncoceutics: Equity Ownership, Membership on an entity's Board of Directors or advisory committees; SentiBio: Equity Ownership; Aptose: Equity Ownership, Membership on an entity's Board of Directors or advisory committees; Oncolyze: Equity Ownership; Jazz Pharma: Consultancy; Reata: Equity Ownership.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal